Infant formula, baby food could soon be safer

Dragana Gordic - stock.adobe.com.
Manufacturers of infant formula and foods currently aren’t required to report contamination—including certain bacteria and heavy metals—to regulators. We think it’s time to change that. Here’s what to know about powdered baby formula, contaminants, and how one state is taking the lead on improving oversight.
New oversight for formula and baby food manufacturers
It’s reasonable to assume that baby foods and formula are safe products. But a pair of issues—company manufacturing processes and mandatory adverse event reporting requirements—have illustrated a gap in regulatory oversight. In the crosshairs of new reform efforts are Cronobacter sakazakii—a foodborne bacterium that sickened four and killed two infants in a 2022 outbreak—and heavy metals, like lead and mercury.
This week, California Governor Gavin Newsom has received CA AB 899, a bill passed by the state Assembly that requires baby food manufacturers who sell or distribute their products in California to test their products for lead, arsenic, cadmium, and mercury—toxic elements that can cause permanent brain damage, cancer, and other serious harm—and make the results of those tests available to the state and the public. The bill awaits his signature to become law.
Also, earlier this year, FDA issued new guidance to powdered infant formula manufacturers it hopes will reduce the risk of cronobacter infections in susceptible infants by improving scientific knowledge, improving oversight over production, and enhancing communication with stakeholders. The agency created the guidance as part of the ongoing response to a 2022 cronobacter outbreak that triggered a recall and temporarily closed one major manufacturing facility—creating a national baby formula shortage during the COVID-19 pandemic. But without the aid of Congress, the agency isn’t authorized to require manufacturers to report cronobacter contaminations.
What is Cronobacter sakazakii?
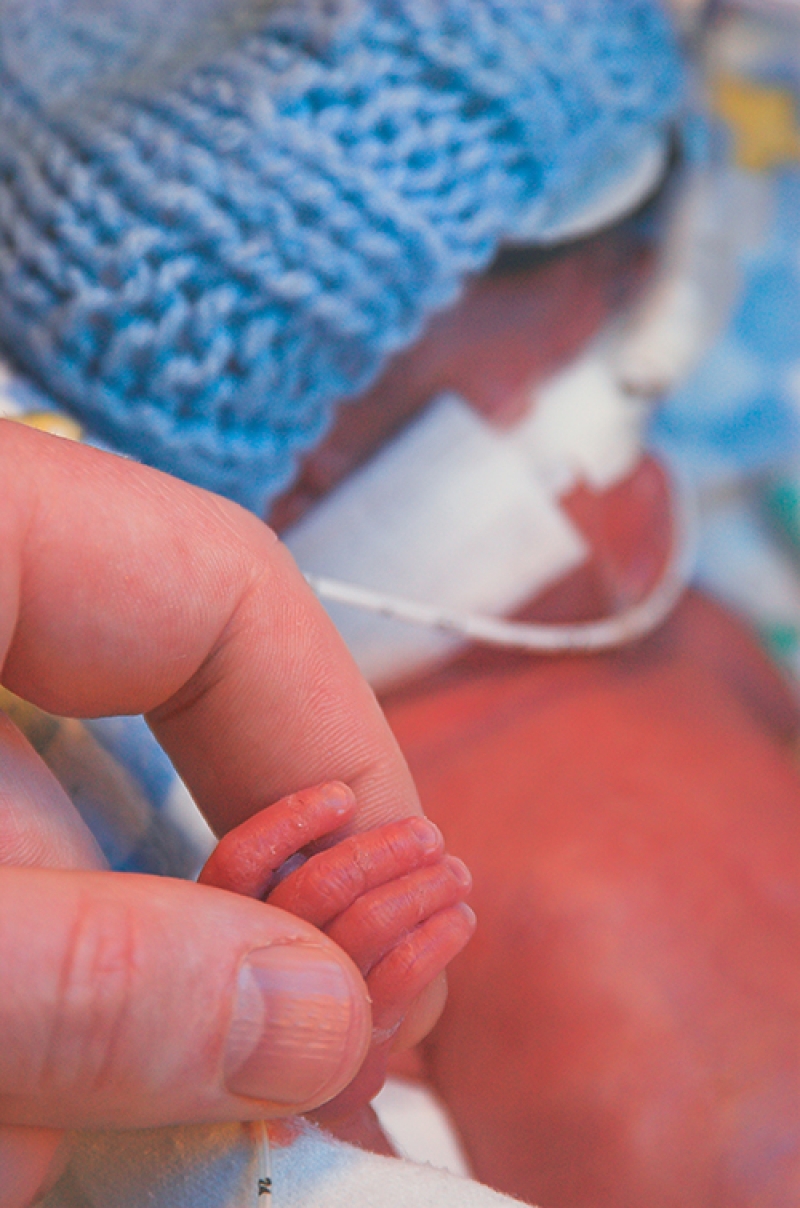
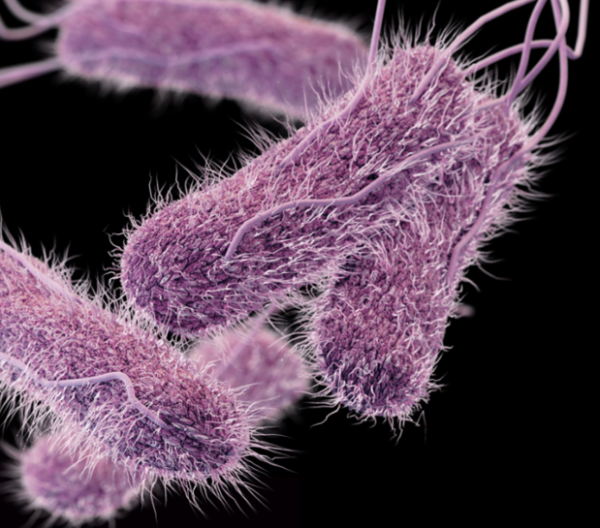
Cronobacter sakazakii occurs naturally and is found in most home settings, where it lives on countertops, sinks, and other hard surfaces. Most people are not susceptible to harmful infections from cronobacter, but for premature or very young infants, the pathogen can cause septicemia (blood poisoning), necrotizing enterocolitis (tissue death in the colon and intestine), and meningitis (swelling of the protective layer around the brain and spinal cord). Cronobacter infection is extremely rare, but is fatal in 40 to 80 percent of cases; survivors generally experience severe neurological disorders, including developmental delays and motor impairment.
Powdered infant formula is considered the primary source of cronobacter in the food supply, but cronobacter testing for non-infant formula products is rare and not required. A 2009 study tested 233 samples of food and environmental products for cronobacter. One powdered infant formula of the 76 tested samples was positive, and 26 of the remaining 28 positive tests were from samples of herbs, spices, teas, and packaged instant foods. (Two positive samples were from household dust.) These non-formula food products are not routinely tested for cronobacter.
What are heavy metals?

Some 20 metal elements, like iron and potassium, are necessary for good health. But others are extremely dangerous—especially for children under the age of five, who are particularly susceptible to environmental toxins. So-called heavy metals are metallic elements , like mercury, cadmium, lead, thallium, arsenic, and chromium, that are toxic in extremely small concentrations. Heavy metals exposure in children has more severe consequences than for adults; the effects include neurological and behavioral disorders, developmental delay and impairment, as well as respiratory problems, cancer, and cardiovascular disease.
Regulators can do better
In August 2023, FDA issued warning letters to three manufacturers, notifying them that agency inspectors detected significant problems with how the plants are addressing cronobacter contamination in their facilities. The good news is that these contaminations were detected early and that no one has reported an illness related to the recalled products. The bad news is that facilities clearly are not doing enough to proactively track down and eliminate cronobacter before it reaches products.
And while California may decide to pick up the slack where FDA has failed to require testing for heavy metals, most Americans don’t live in California. We need federal oversight with mandatory reporting for heavy metal contaminants and pathogens that pose serious risks to the lifelong health of children and infants.
Baby formula, of all foods, should be safe. But under current federal law, baby food manufacturers are not required to test their products for heavy metals; when they do so voluntarily, they are not required to share the results with federal or state authorities—or the public.
Cronobacter has only recently been added to the list of 120 diseases the CDC tracks in its National Notifiable Diseases Surveillance System. Mandatory notification begins in 2024.
How CSPI is working to help
In May 2023, CSPI Executive Director Dr. Peter Lurie, speaking to CNN, applauded the new cronobacter guidance, saying, “I think it’s a necessary step. It is difficult to prevent diseases that you can’t count.” Lurie also stressed the need for mandatory reporting from manufacturers, which requires authorization by Congress, and expanding testing for cronobacter to other food products, like dry herbs, teas, and powdered instant food products, which are not routinely tested for the bacterium but are potential sources of food supply contamination. "I think we have a lot ot learn there," Lurie said.
For California residents, CSPI urges Gov. Newsom to sign California AB 899 into law and require formula-makers to test for and report heavy metal contaminations of infant products sold in the state.
What you can do
Parents and caregivers of infants and children who consume powdered formula should stay abreast of FDA recall and food safety alerts and follow the CDC guidelines for avoiding cronobacter. These include breastfeeding when possible; cleaning and storing bottles safely; keeping the hard surfaces of your home clean; handwashing; and proper handling and storage of powdered formula.
We urge Gov. Newsom to sign CA AB 899 into law, and protect the people of California until FDA takes action on heavy metal contamination. You can support our action by sending a letter to Gov. Newsom urging him to sign AB 899, as well as AB 418, which bans four dangerous food additives.
Regulatory News
Bold USDA move on Salmonella in poultry takes a new approach to reduce rates of infection in people
Food Safety
USDA sets first added sugars limit on school breakfasts, lunches
Government Accountability

School meals get an upgrade: What to expect going forward
Government Accountability

What to know about the food additives now banned in California
Food Additives

Illinois moves to ban Red 3 and other harmful food additives
Government Accountability


