Prostate cancer: a new treatment era

Syda Productions - stock.adobe.com.
"Ten years ago, everyone was getting radical treatment for prostate cancer,” says Laurence Klotz, chairman of the World Urological Oncology Federation. “Everybody. Even low-risk patients. Then the world changed.”
Now researchers know far more about who to treat and who to keep under “active surveillance.” And now urologists can use new techniques—from MRI scans to tests for genetic aberrations—to help men decide what to do.
“It’s a new era,” says Klotz. “This whole area is changing rapidly.”
To screen or not to screen...
“I got diagnosed with prostate cancer Friday, June 13th, 2014,” wrote the actor Ben Stiller in 2016 on the website Medium.
“On September 17th of that year I got a test back telling me I was cancer free. The three months in between were a crazy roller coaster ride with which about 180,000 men a year in America can identify.”
That was 2016. By 2017, the number of men diagnosed with prostate cancer had dropped to an estimated 160,000. Is that good news?
“Normally, we would celebrate a decline in cancer incidence rates,” wrote Meir Stampfer and others in a recent issue of JAMA Oncology.1
But prostate cancer is different.
“Many, perhaps most, prostate cancers never cause harm because they are indolent,” says Stampfer, professor of epidemiology and nutrition at the Harvard T.H. Chan School of Public Health.
In fact, autopsy studies find undiagnosed prostate cancer in more than a third of Caucasian and half of African-American men aged 70 to 79.2
“That tells you right off the bat that most prostate cancer is harmless,” says Stampfer.

But finding and treating those cancers does cause harm.
For every 1,000 men aged 55 to 69 who are screened, 100 are diagnosed with prostate cancer. And treatment—surgery or radiation—saves only one or two lives. (See “PSA: By the Numbers.")
Meanwhile, 60 of the 100 experience impotence or urinary incontinence. And that doesn’t include the anxiety or pain felt by the 120 who undergo a biopsy that finds no cancer.3
In 2012, that led the U.S. Preventive Services Task Force (USPSTF) to recommend against screening for high blood levels of PSA (prostate-specific antigen), which had been the main screening tool since 1994.
“The main driver was the huge overtreatment of prostate cancer,” says Stampfer. “We had lots of men getting surgery and radiation for slow-growing tumors that most likely never would have caused any harm if they weren’t diagnosed.”
But no screening also has a downside.
“There’s clearly harm caused by overtreatment, but it doesn’t make sense to stop PSA testing completely, because some tumors are aggressive, and we can do something about them,” says Stampfer. “Prostate cancer is still the third leading cause of cancer death in U.S. men.”
In April 2017, the USPSTF issued a new recommendation: Men aged 55 to 69 should talk to their doctor about a PSA test. (Older men can still skip the test, said the USPSTF.)4
“That’s better,” says Stampfer. But he’s worried that too few men will get tested.
“A few years after screening started to take hold, the incidence of metastatic cancers at the time of diagnosis fell dramatically, because those cancers were being picked up earlier,” says Stampfer. “And we saw a big drop in prostate cancer mortality.”
But the decline has tapered off.
“Death rates have been that, and my fear is that they’ll start going back up,” says Stampfer. “PSA testing lets you diagnose the disease about 10 years early,” so deaths may not rise until 2022.
“Instead of putting our heads in the sand by not screening, a more reasonable solution is to do targeted PSA testing,” he argues.
For example, a single PSA test may be enough for many men. “If your PSA is 1 or less at age 60—that’s true for about half of all men—we can say ‘Good news, you’re not going to die of prostate cancer,’ and you’re done,” says Stampfer.
Men who are African-American or who have a family history of prostate cancer should get screened at age 45, he suggests.

But it makes no sense to screen men whose life expectancy is less than 15 years, he notes. “In the early days of testing, my 90-year-old father had a PSA test. That’s just silly.”
What’s more, doctors are getting better at predicting which men with high PSA scores need a biopsy, and which men with cancers need to be treated.
A new era
Five years ago, many doctors would do a biopsy on any man with a PSA over 4.
“Now, unless it’s really high, the doctor might say ‘Okay, come back in three or six months,’” says Stampfer. “A high PSA could be due to a prostate infection or too much bike riding or just a fluke.”
And doctors have new tests to predict the odds of whether you have cancer and whether it’s aggressive enough to treat.
“This whole area is changing rapidly,” says Laurence Klotz, past chief of urology at the Sunnybrook Health Sciences Centre in Toronto.
For starters, instead of going straight from a high PSA to a biopsy, urologists can order a second test—like a 4K score, Phi, or Select MDx—to look at types of PSA or other markers in blood or urine.
“These tests clearly identify a group of patients who are at higher risk,” says Klotz, who chairs the World Urological Oncology Federation and the Canadian Urology Research Consortium.
For some men, the next step may be an MRI (magnetic resonance imaging). “The MRI is a game changer,” says Klotz.
A key question: Can you skip a biopsy if an MRI finds no cancer?
“In our large national study, MRI didn’t work as well as we had hoped,” says Klotz. “Most patients still needed a biopsy even if the MRI found no cancer.”
So for now, the answer depends on how much of a risk to take.
“If a man’s only risk factor is a slightly elevated PSA, the MRI can probably replace the biopsy,” says Klotz. “But a high-risk patient is going to need a biopsy even if the MRI shows no cancer.”
A biopsy reveals not just who has cancer, but the tumor’s Gleason score. And that’s still the key to deciding whom to treat and whom to keep on “active surveillance.” That means men periodically get a PSA, a biopsy, or other tests, and get treated if necessary.5
Gleason & beyond
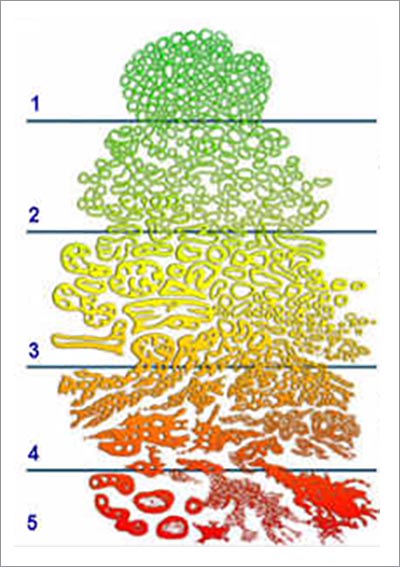
When you get a biopsy, a pathologist gives the prostate cells a Gleason grade, which ranges from 1 (most normal) to 5 (most abnormal).
Your Gleason score is made up of two Gleason grades: the most common + the most abnormal. So if most of your cells are Gleason grade 3, but a few are grade 4, your score is 3 + 4. But if most are grade 4, and a few are grade 3, your score is 4 + 3.
And 3 versus 4 can be the difference between night and day.
“Most Gleason grade 3 cells look normal,” says Klotz. “They lack most of the hallmarks of cancer, and the DNA in about 90 percent of the samples has no molecular aberrations.”
In contrast, “Gleason grade 4 cells look like cancer,” he says. “The DNA has deleted tumor suppressor genes and other aberrations. It’s a real cancer.”
His Toronto Surveillance Cohort has tracked nearly 1,000 patients for up to 20 years.6 They got periodic biopsies and PSA tests, and they were treated if their Gleasons got worse.
“Having a baseline PSA above 10 didn’t have much impact,” says Klotz. Nor did a rapid rise in PSA.
What mattered: having any Gleason 4.
“Even in the most-favorable 3 + 4 group, about 20 percent metastasized by 15 years despite careful monitoring,” says Klotz.
“That’s too high, so we now say either treat the patient or be very, very cautious.” (Anyone with a less-favorable Gleason 4 + 3 gets treated.)
In contrast, Johns Hopkins University only accepted men at very low risk into its active surveillance cohort. (For example, they had no Gleason grade 4 and couldn’t have Gleason grade 3 in more than two samples from one biopsy.) Of 1,300 men, only five had metastases after 15 years. Another 47 died of other causes.7
“Now Hopkins pretty much offers surveillance to nearly all men with a Gleason 3 + 3 score,” says Klotz.
And urologists now classify Gleason 3 + 3 as Grade Group 1, not just because it’s less risky, but because it sounds less advanced. (Gleason 3 + 4 is Group 2, and Gleason 4 + 3 is Group 3.)
Of course, doctors consider other factors before advising each patient.
Nevertheless, says Klotz, “we can now offer active surveillance to a third to 40 percent of newly diagnosed patients.”
New gene tests
“We have no problem putting patients on surveillance in Canada,” says Klotz. “Men accept it and don’t seem to have anxiety about it.”
But some men, he says, especially “south of the border”—that is, in the United States—are anxious or get infected with anxiety by their physicians.
For those men, an MRI and new genetic tests can help. What’s more, they can help ensure that a biopsy didn’t miss a Gleason 4.
“That is the Achilles’ heel of this field,” says Klotz. It happens in 25 to 30 percent of patients with Gleason 3 + 3. In contrast, he says, “true progression from a Gleason 3 to a 4 or a 5 occurs in only 1 to 2 percent of cases per year.”
So far, three new tests—Decipher, Oncotype DX, and Prolaris—are being used to test prostate tissue genes for signs that the cancer is aggressive.
“The tests can predict patients who are more at risk for dying of prostate cancer based on the pattern of genetic expression,” says Klotz. The tests are unnecessary for most low-risk patients.
“We mainly use them where you have a discrepancy,” says Klotz. “For example, you have a patient who has a little bit of Gleason pattern 4, but most of the disease is the more favorable Gleason 3. The MRI shows no cancer. The PSA is favorable. Does that patient really need surgery or radiation? The tests are quite useful in those kinds of borderline situations.”

Some genetic tests can also detect if men have genes like BRCA1 or BRCA2.
“These patients don’t repair mistakes in their DNA, so they accumulate a massive number of mutations rapidly,” says Klotz. “These men should not be on surveillance because they deteriorate rapidly.”
Men with BRCA1 or 2 make up about 3 percent of the population but closer to 10 to 15 percent of men with prostate cancer, he adds. Most have a family history of breast or ovarian cancer.
“It’s not a stretch to say that within a year or so, we’re going to be screening every surveillance candidate for those mutations,” adds Klotz.
That’s the future, he says.
“With genetic testing, we’re going to move to an era where there’s no gray zone about who to treat and who to follow. You’ll be able to predict with significant accuracy what’s going to happen.”
Lower your risk
Harvard’s Meir Stampfer isn’t looking for links to a higher risk of all prostate cancers.
“We’re interested in what raises the risk of lethal prostate cancer, not indolent disease,” he says.
Of the risk factors that you can change, “smoking within the previous 10 years is one of the big ones,” says Stampfer.
The evidence on others is growing but still limited:
Exercise. “We found about a 60 percent decreased risk of lethal prostate cancer in men with prostate cancer who were doing at least three hours of vigorous activity per week,” says Stacey Kenfield, associate professor of urology at the University of California, San Francisco.8
Results were similar in healthy men over 65.9 And in another study of men with prostate cancer, even those who walked briskly for three hours a week had a lower risk of progression (for example, a rising PSA).10
“Brisk walking could be vigorous activity for some older men,” say Kenfield. “If your heart rate is elevated, you’re breathing rapidly, and you’d rate your exertion level as ‘hard to very hard,’ you’re doing vigorous exercise.”
Weight. Men who are obese have a higher risk of fatal prostate cancer.
“Higher levels of insulin-like growth factor, insulin, and inflammation due to obesity may promote prostate tumors,” says Kenfield.11
Cooked tomatoes. In a recent study, men who consumed tomato sauce at least twice a week had a 45 percent lower risk of prostate tumors with a common gene abnormality caused by poorly repaired DNA strand breaks.12 The lycopene in tomatoes may protect the prostate.
“I wouldn’t say it’s proven, but I recommend cooked tomatoes with some oil, because lycopene is fat soluble,” says Stampfer. “Raw tomato won’t raise your blood levels of lycopene much.”
Fish. In a study of roughly 2,100 men with prostate cancer, those who ate fish at least five times a week were half as likely to die of prostate cancer than those who ate fish less than once a week.13
“Fatty fish is an excellent source of omega-3 fatty acids,” says Kenfield. Those fats may slow the growth of cancer cells.

Dairy. In a study of 1,334 men with prostate cancer, “we found that those who drank more than four servings of whole milk a week had a 73 percent increased risk of prostate cancer recurrence than men who had no more than three servings a month,” says Kenfield.
“Low-fat milk and other dairy foods were not associated with recurrence.”14
Men who consumed more saturated fat after diagnosis also had a higher risk of dying of prostate cancer.15
Calcium & phosphorus. Men who get more than 2,000 milligrams of calcium a day—from food plus supplements—have a higher risk of lethal prostate cancer.
But the link may be due to phosphorus, which is found along with calcium in dairy foods, as well as in thousands of foods with phosphate-containing additives.16 Stay tuned.
Selenium & vitamin E. The SELECT trial gave 35,000 men selenium (200 micrograms a day) or vitamin E (400 IU a day), both, or a placebo for five years. It didn’t turn out as researchers had hoped.
Among men who entered the trial with high levels of selenium, those who took selenium had a higher risk of high-grade prostate cancer. And among men who started with low selenium levels, those who took vitamin E had a higher risk of high-grade prostate cancer.17
Results like those make Kenfield wary of any supplements for the prostate.
“If your vitamin D level is low, it’s completely appropriate to take a supplement,” she says. But if you’re not deficient, “there’s no good evidence supporting any single supplement after diagnosis.”
On the other hand, two drugs hold promise. “There’s a lot of data that statins slow the progression of prostate cancer,” says Klotz. “And men taking metformin for diabetes have a lower rate of prostate cancer mortality.”
But we don’t yet have rock-solid evidence from trials pitting each drug against a placebo, he notes. A trial on metformin is under way.
Other studies are also in the works.
“By my count, there are about 15 trials looking at various lifestyle interventions in men on surveillance,” says Klotz. “We’ll have data from them probably in four or five years.”
Until then, “all the epidemiologic data says that if a diet is good for your heart, it’s good for your prostate,” says Klotz.
“That means not too much animal fat, not too much red meat, lots of fruits and vegetables, and avoid obesity.”
Want more info?
For more about biomarkers that help decide who needs a biopsy or treatment, go to pcmarkers.com.
To estimate your risk, go to prostatecancer-riskcalculator.com.
To watch a 2017 scientific presentation by Dr. Klotz, go to grandroundsinurology.com/active-surveillance-resultsimplications-studies.
1JAMA Oncol. 3: 1623, 2017.
2Int. J. Cancer 137: 2795, 2015.
3 screeningforprostatecancer.org/get-the-facts.
4 uspreventiveservicestaskforce.org/Page/Document/ draft-recommendation-statement/prostatecancer- screening1.
5JAMA 318: 2152, 2017.
6J. Clin. Oncol. 33: 272, 2014.
7J. Clin. Oncol. 33: 3379, 2015.
8J. Clin. Oncol. 29: 726, 2011.
9Arch. Intern. Med. 165: 1005, 2005.
10Cancer Res. 71: 3889, 2011.
11J. Natl. Cancer Inst. 109: djw225, 2017.
12Am. J. Clin. Nutr. 103: 851, 2016.
13Am. J. Clin. Nutr. 88: 1297, 2008.
14Prostate 78: 32, 2018.
15Cancer Causes Control 26: 1117, 2015.
16Am. J. Clin. Nutr. 101: 173, 2015.
17J. Natl. Cancer Inst. 106: djt456, 2014.
Photos: Syda Productions/stock.adobe.com (man), Grosso/stock.adobe.com (fish).
Graphics: Int J. Cancer 2015. doi:10.1002/ijc.29408 (autopsy studies), adapted from U.S. Preventive Services Task Force, screeningforprostatecancer.org/get-the-facts (PSA screening), SEER Program, National Cancer Institute, seer.cancer.gov/statfacts/html/prost.html (death rates).
Tags
Topics

